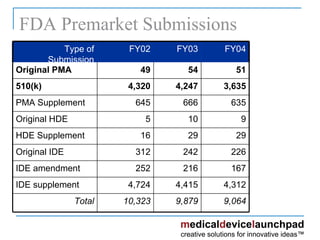
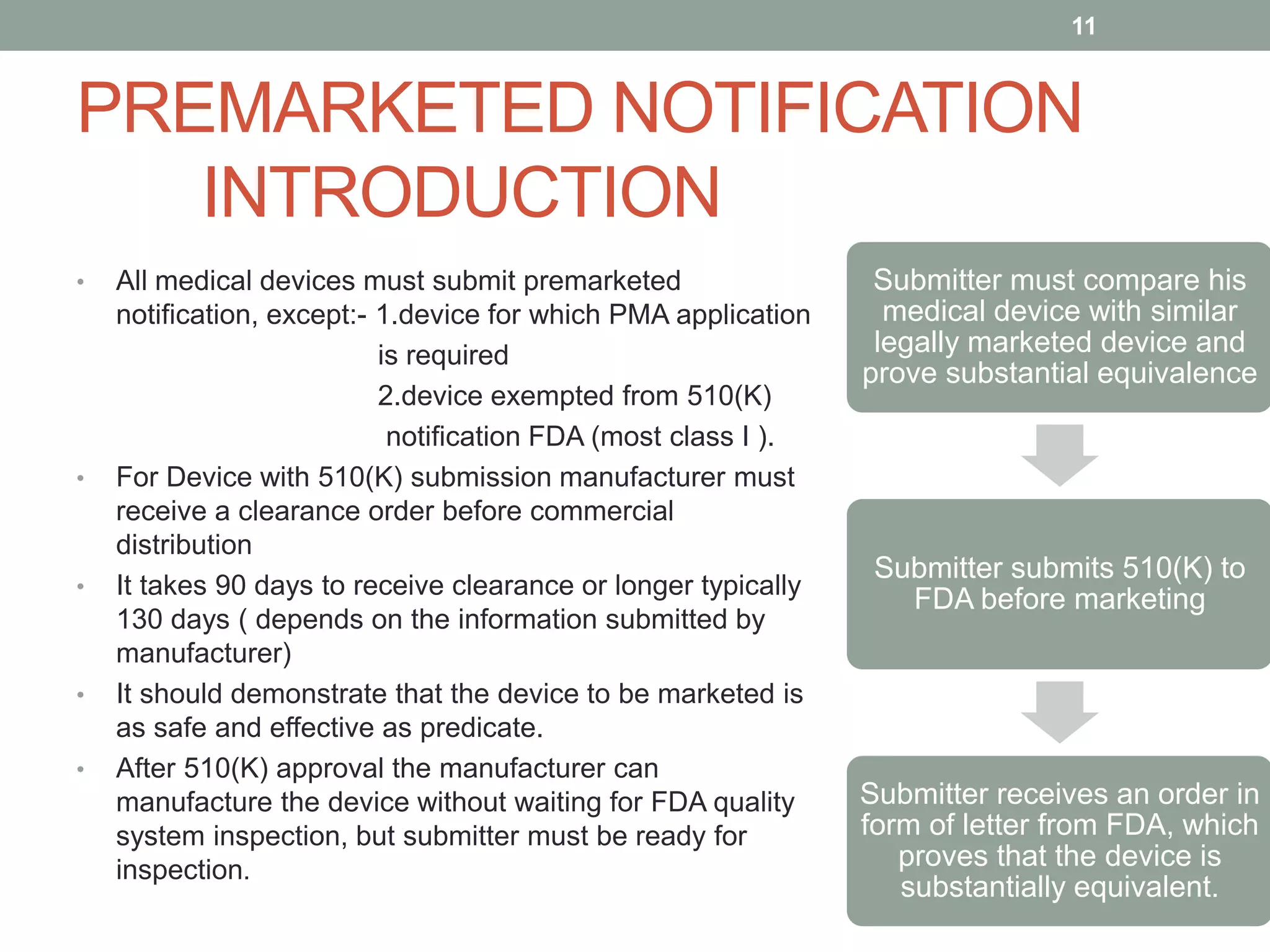
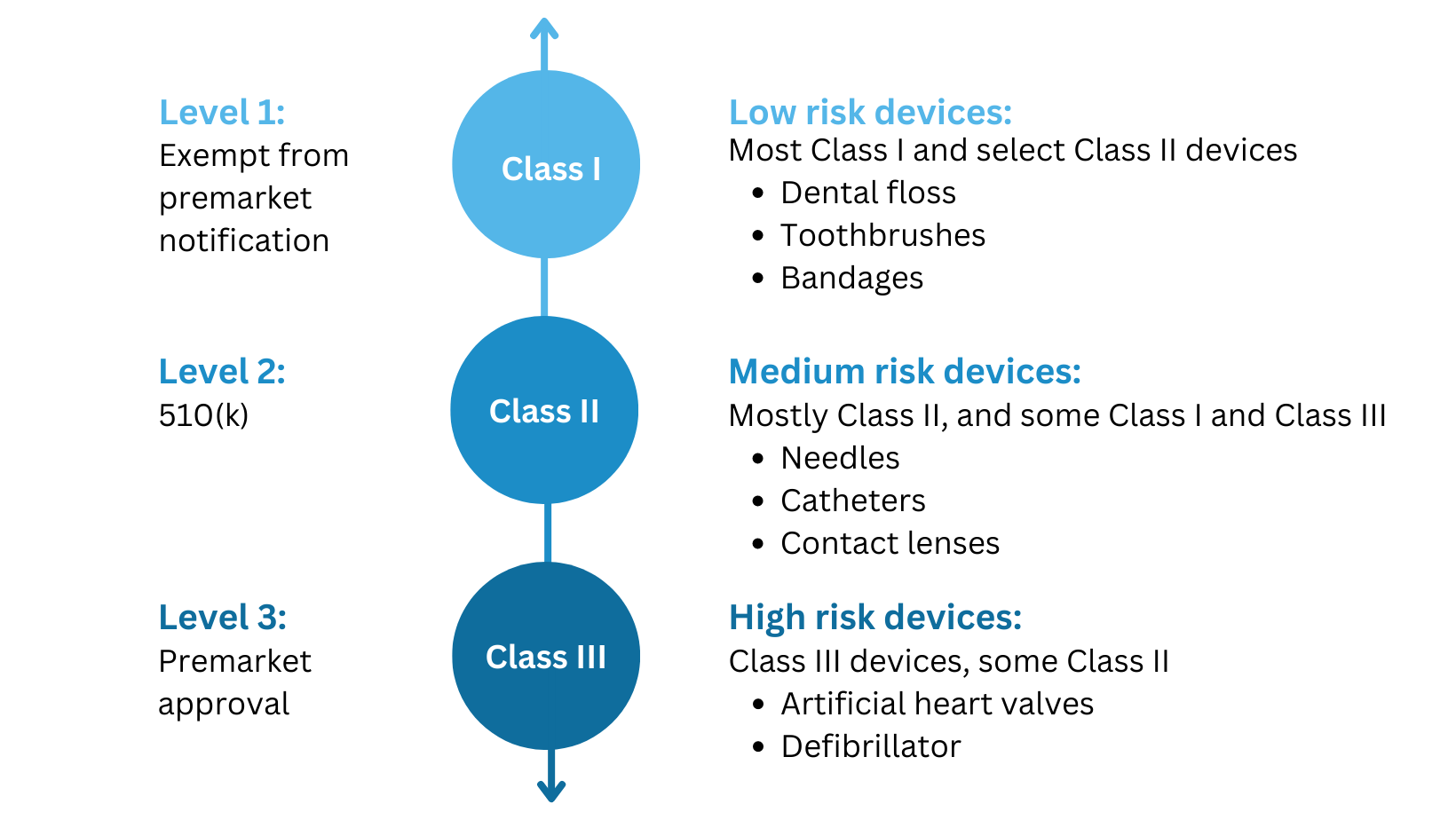
Embark on an remarkable adventure with our 2021 draft fda guidance - content of premarket submissions for device collection featuring extensive collections of captivating images. showcasing the dynamic nature of computer, digital, and electronic. ideal for travel bloggers and adventure photographers. Discover high-resolution 2021 draft fda guidance - content of premarket submissions for device images optimized for various applications. Suitable for various applications including web design, social media, personal projects, and digital content creation All 2021 draft fda guidance - content of premarket submissions for device images are available in high resolution with professional-grade quality, optimized for both digital and print applications, and include comprehensive metadata for easy organization and usage. Our 2021 draft fda guidance - content of premarket submissions for device gallery offers diverse visual resources to bring your ideas to life. Whether for commercial projects or personal use, our 2021 draft fda guidance - content of premarket submissions for device collection delivers consistent excellence. Each image in our 2021 draft fda guidance - content of premarket submissions for device gallery undergoes rigorous quality assessment before inclusion. Regular updates keep the 2021 draft fda guidance - content of premarket submissions for device collection current with contemporary trends and styles. Reliable customer support ensures smooth experience throughout the 2021 draft fda guidance - content of premarket submissions for device selection process.
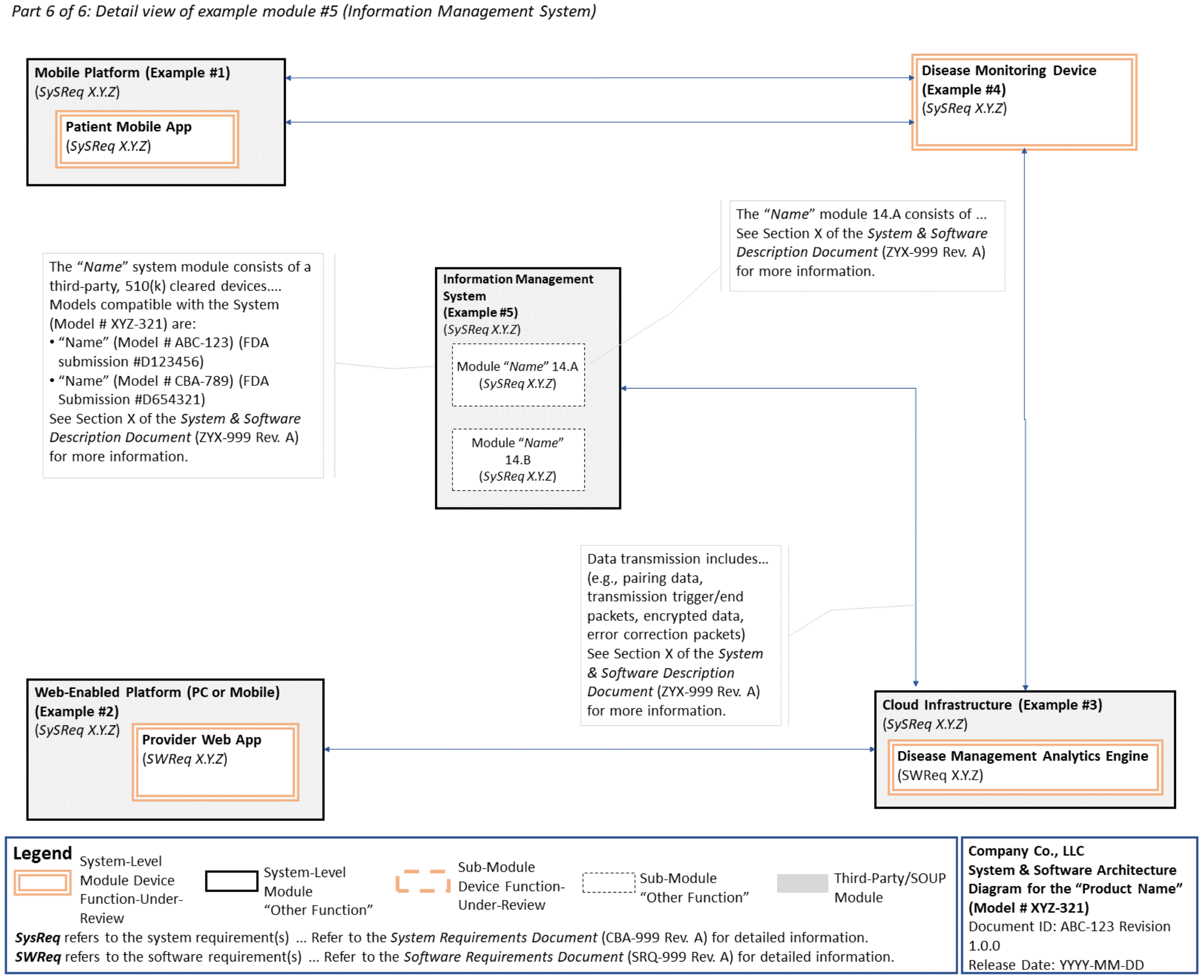

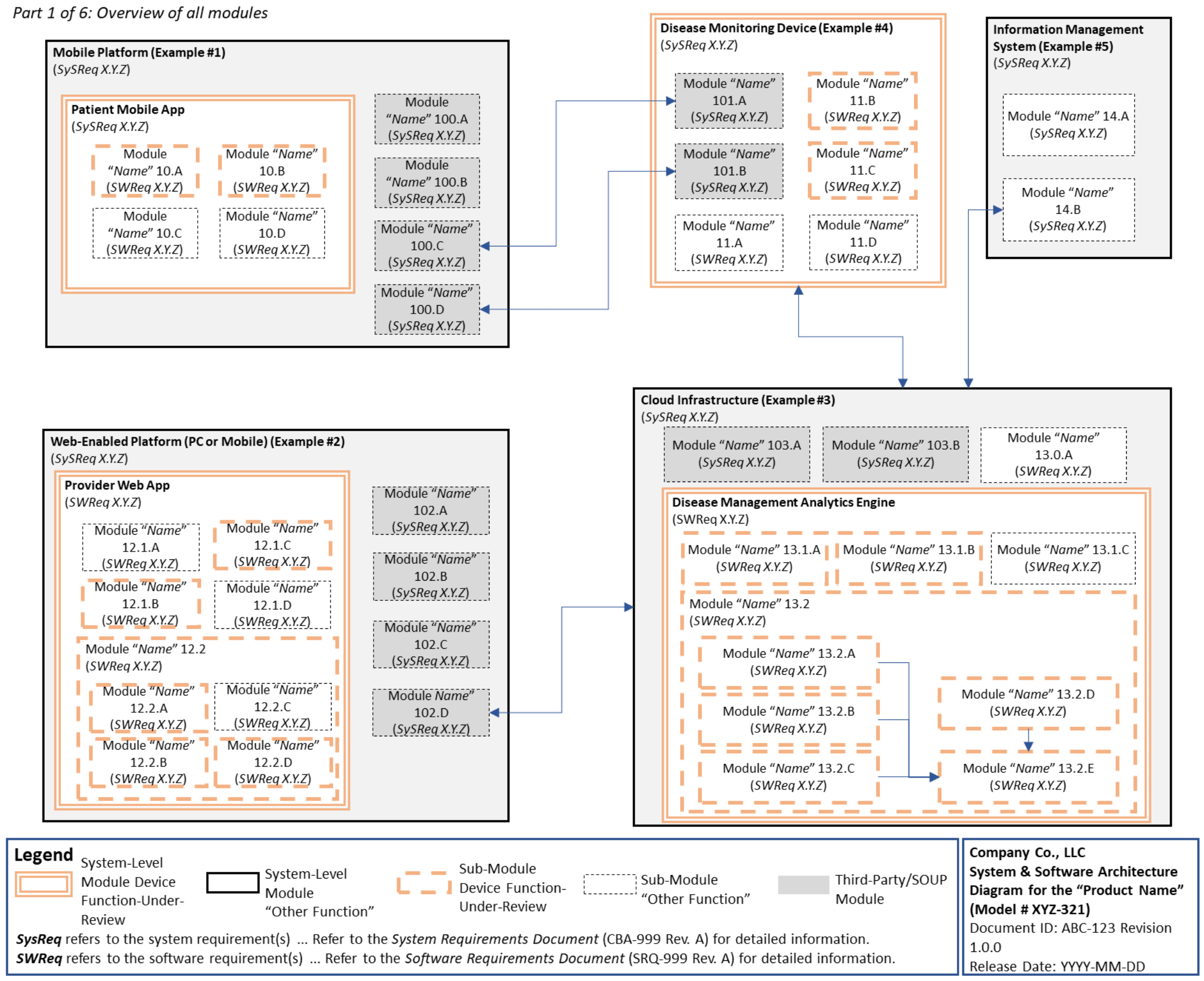














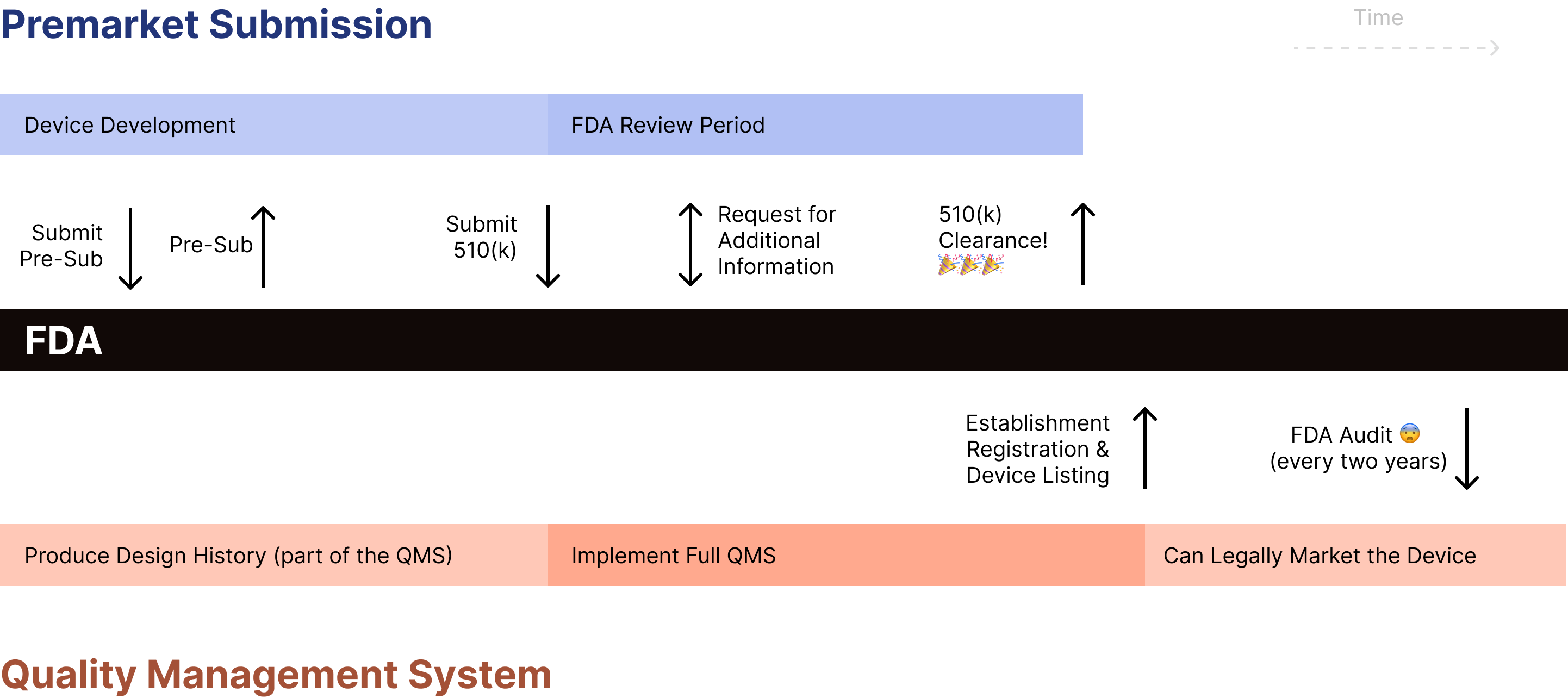

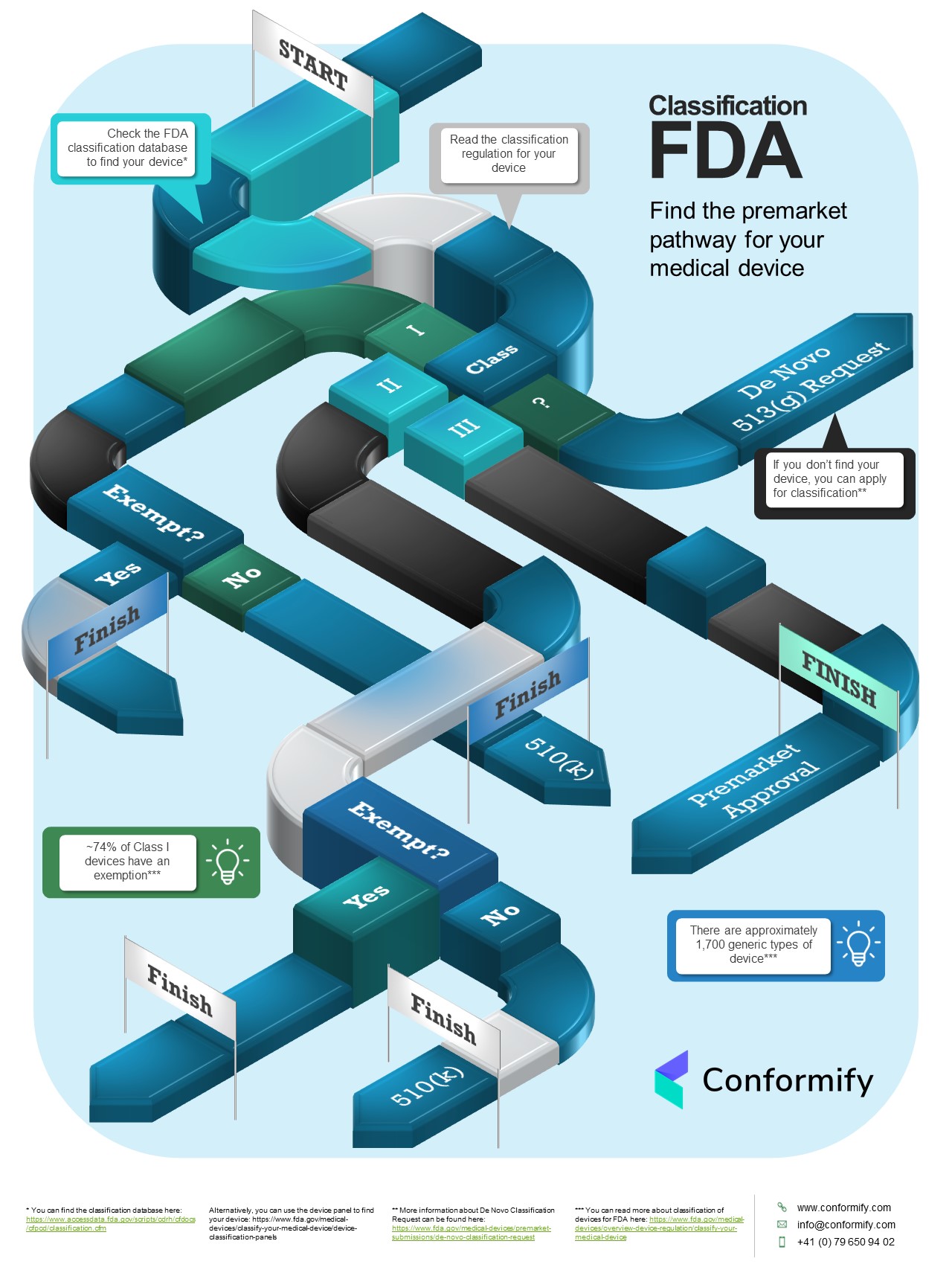

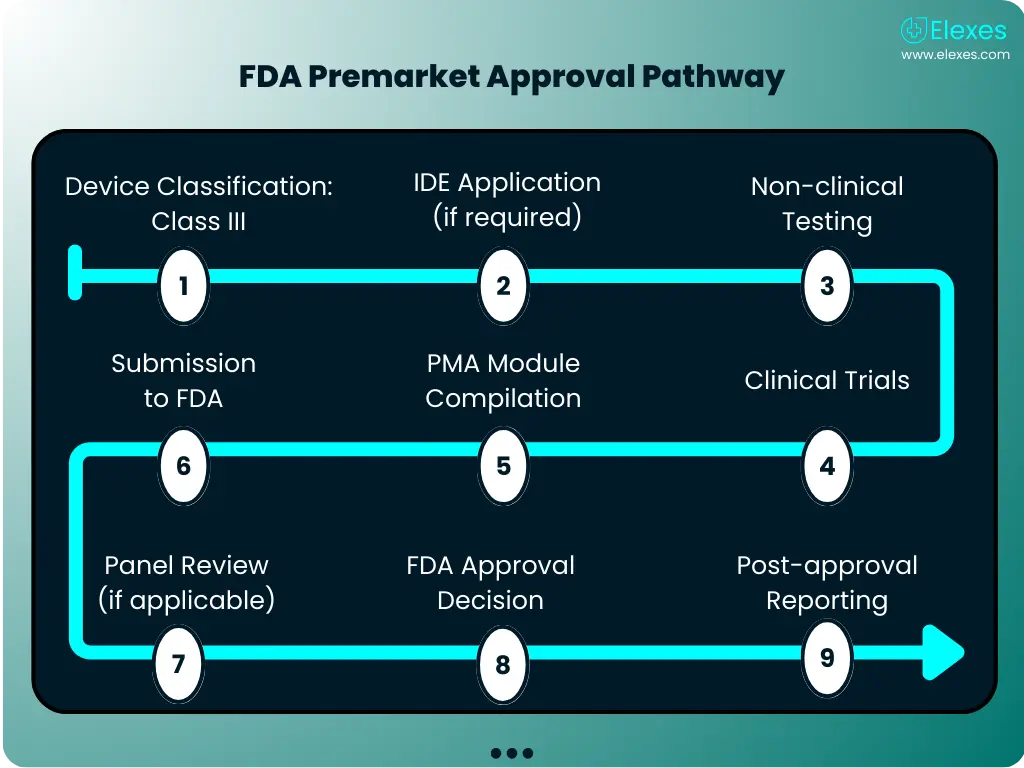




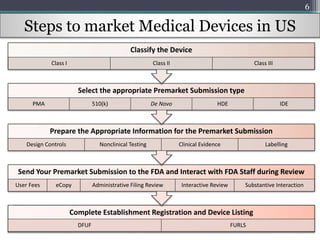
-chcecklist-complete-guide-fda-510k-submissions-ppg.png)




%20-%20Process%20and%20Preparation.jpg)




























.png?width=800&height=400&name=How%20To%20Use%20FDA%E2%80%99s%20Guidance%20on%20Post-Approval%20Studies%20During%20Premarket%20Approval%20(new).png)