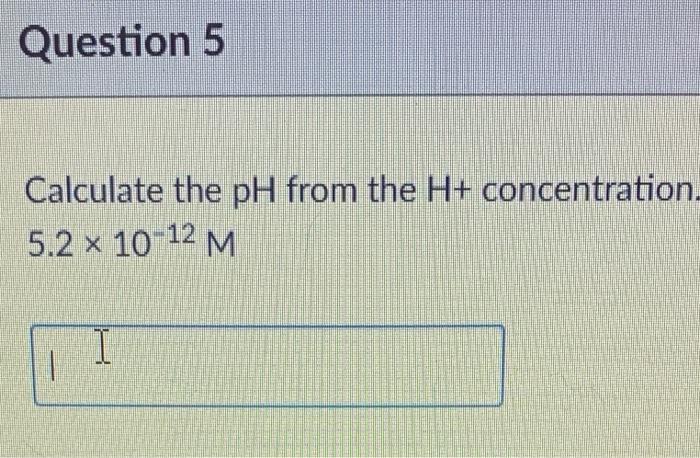
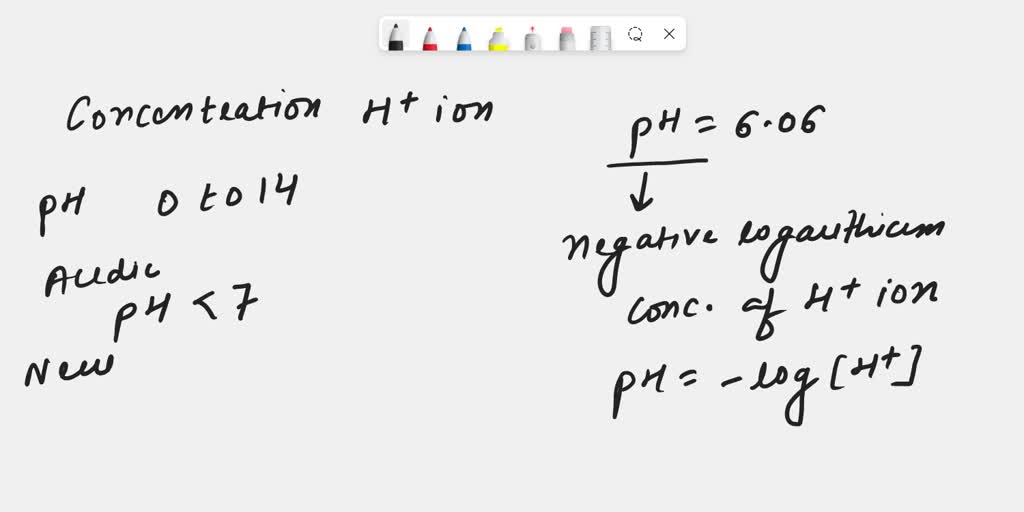
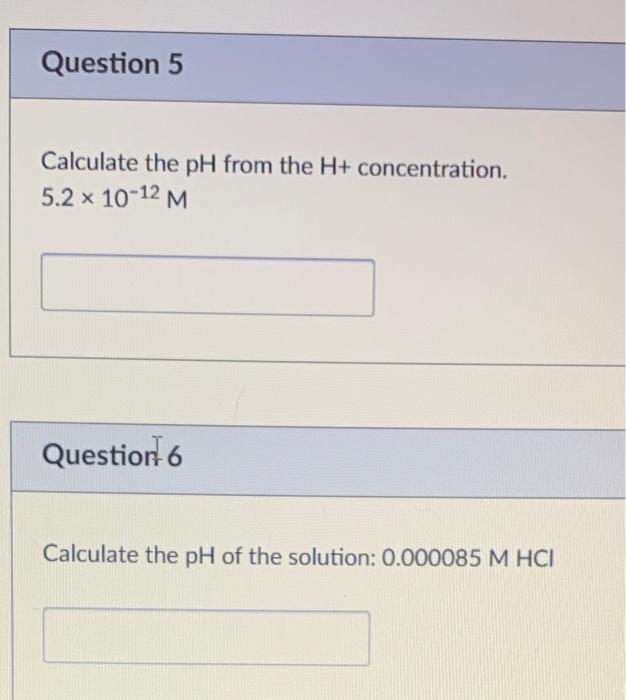
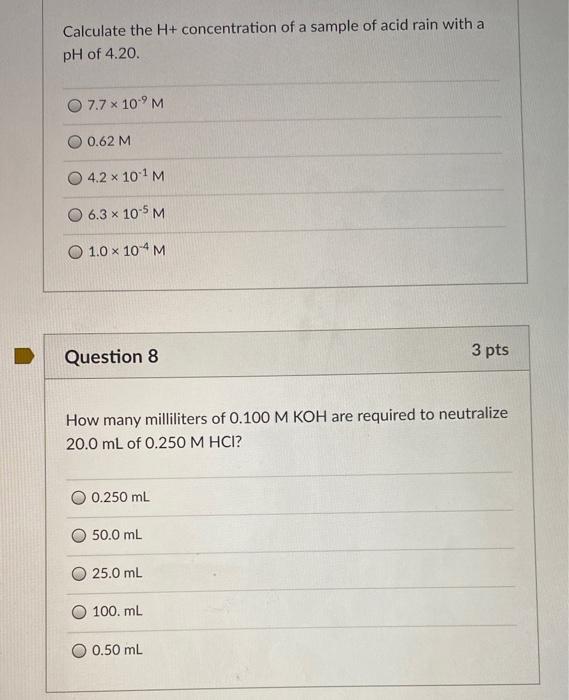
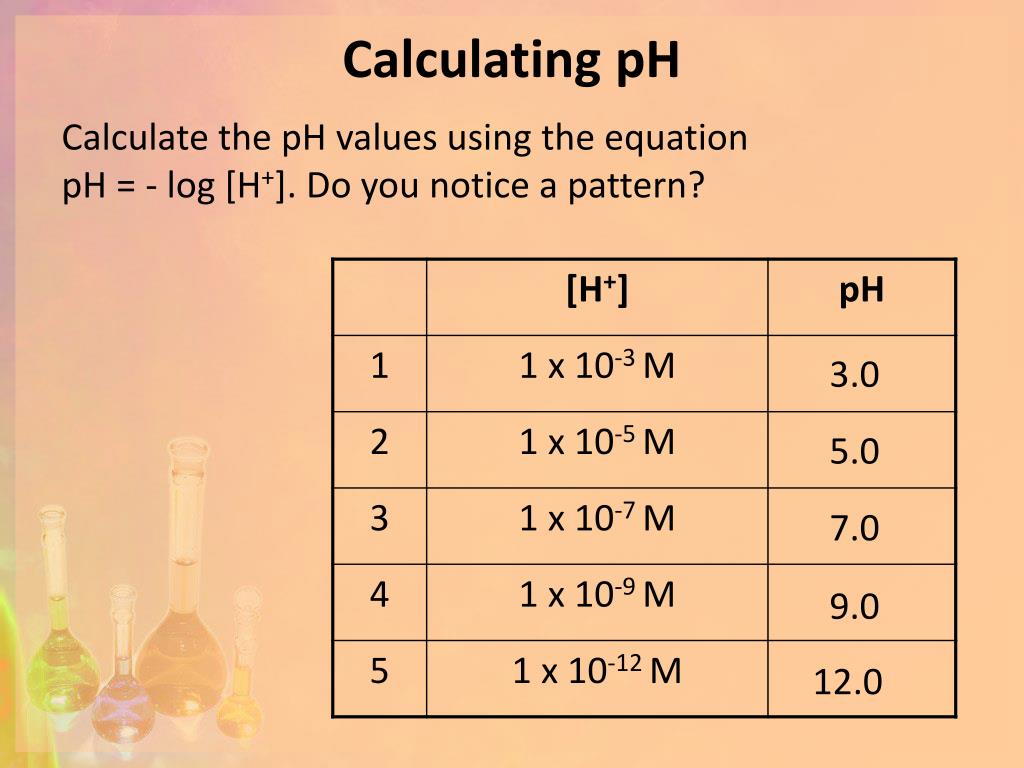
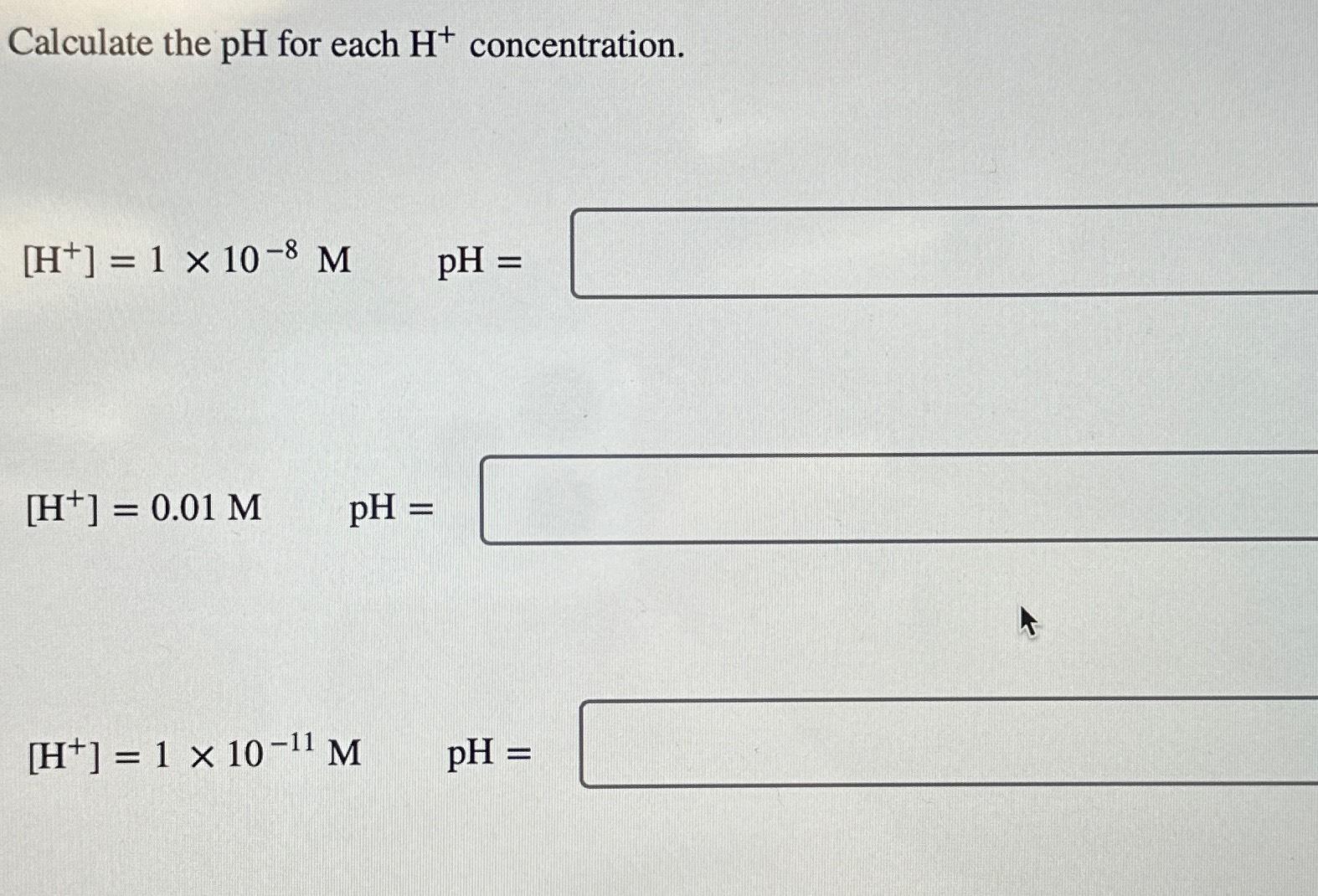
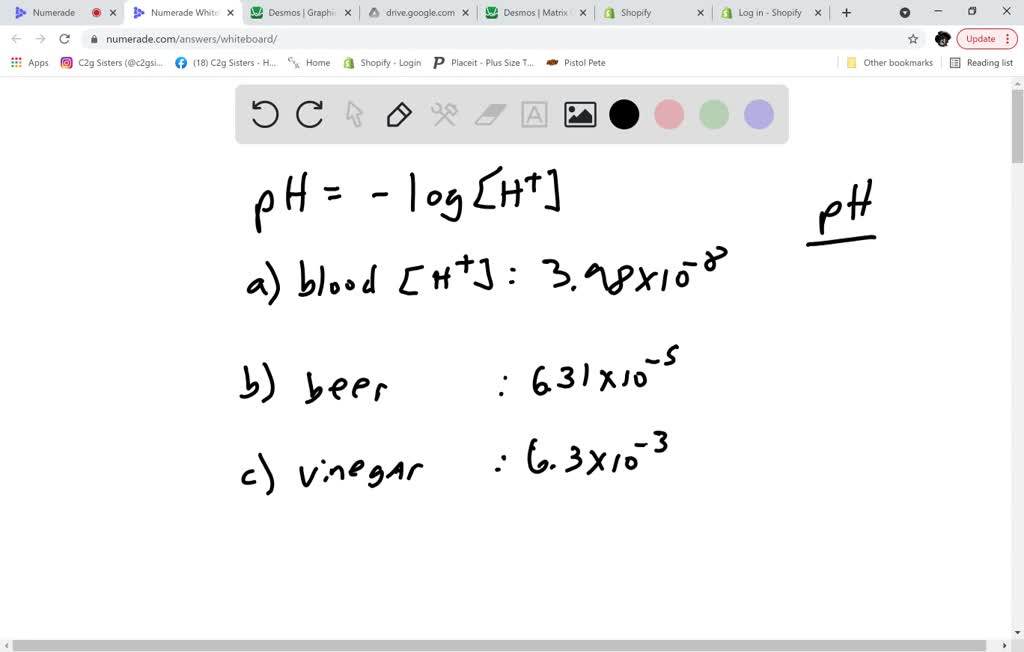
Calculating Ph From H Concentration
Savor the flavor with our stunning culinary Calculating Ph From H Concentration collection of vast arrays of appetizing images. appetizingly showcasing photography, images, and pictures. perfect for restaurant marketing and menus. Each Calculating Ph From H Concentration image is carefully selected for superior visual impact and professional quality. Suitable for various applications including web design, social media, personal projects, and digital content creation All Calculating Ph From H Concentration images are available in high resolution with professional-grade quality, optimized for both digital and print applications, and include comprehensive metadata for easy organization and usage. Our Calculating Ph From H Concentration gallery offers diverse visual resources to bring your ideas to life. Diverse style options within the Calculating Ph From H Concentration collection suit various aesthetic preferences. Whether for commercial projects or personal use, our Calculating Ph From H Concentration collection delivers consistent excellence. Each image in our Calculating Ph From H Concentration gallery undergoes rigorous quality assessment before inclusion. Regular updates keep the Calculating Ph From H Concentration collection current with contemporary trends and styles. The Calculating Ph From H Concentration collection represents years of careful curation and professional standards. Reliable customer support ensures smooth experience throughout the Calculating Ph From H Concentration selection process.




![Calculating pH from [OH-] hydroxide Concentration - CLEAR & SIMPLE ...](https://i.ytimg.com/vi/gn1CgBzShps/maxresdefault.jpg)

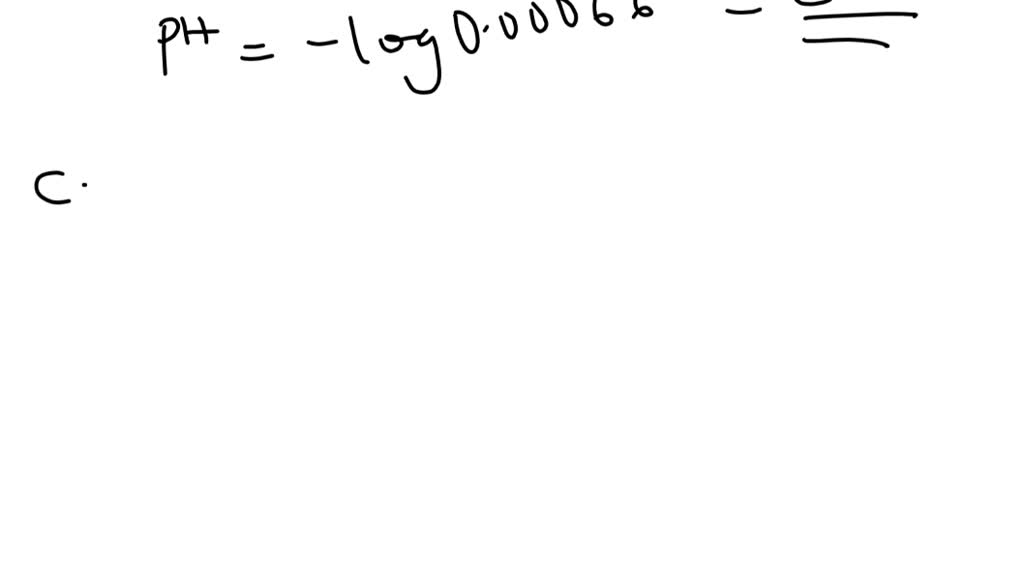







![How to Calculate hydrogen ion concentration [H+] FROM pH | pH ...](https://i.ytimg.com/vi/5IEb44Y_W28/maxresdefault.jpg)



![Solved Calculate the pH for each H+ concentration. [H+] = 1 | Chegg.com](https://media.cheggcdn.com/study/a68/a68e86e2-b729-405b-a8cd-1c779e01cb5f/image.png)
![Solved Calculate the pH for each H+ concentration. [H+] = 1 | Chegg.com](https://media.cheggcdn.com/study/0f3/0f3fbd2c-fe2d-4d56-b457-94e62686c896/image)
![Solved Calculate the pH for each H+ concentration. [H+] = 1 | Chegg.com](https://media.cheggcdn.com/study/40f/40fa63cf-d973-4b1c-a0d5-08f40420eacd/image)

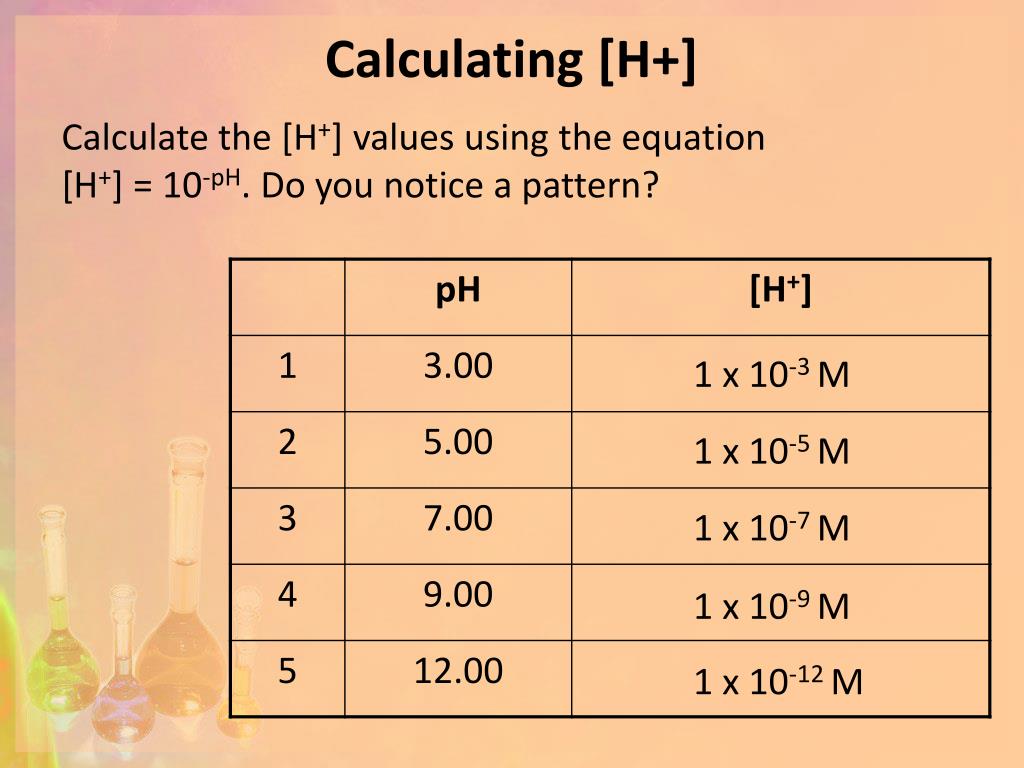


![Solved Calculate pH for each H concentration a) [H+]=1x10-8 | Chegg.com](https://media.cheggcdn.com/media/57f/57f9b328-01cd-4663-93c9-4a104cb44113/image)

![Solved Calculate the pH for each H+ concentration. [H+] = 1 | Chegg.com](https://media.cheggcdn.com/media/3e9/3e9c8566-7c50-490a-8652-32a153b3f12d/phpGDsHTZ.png)


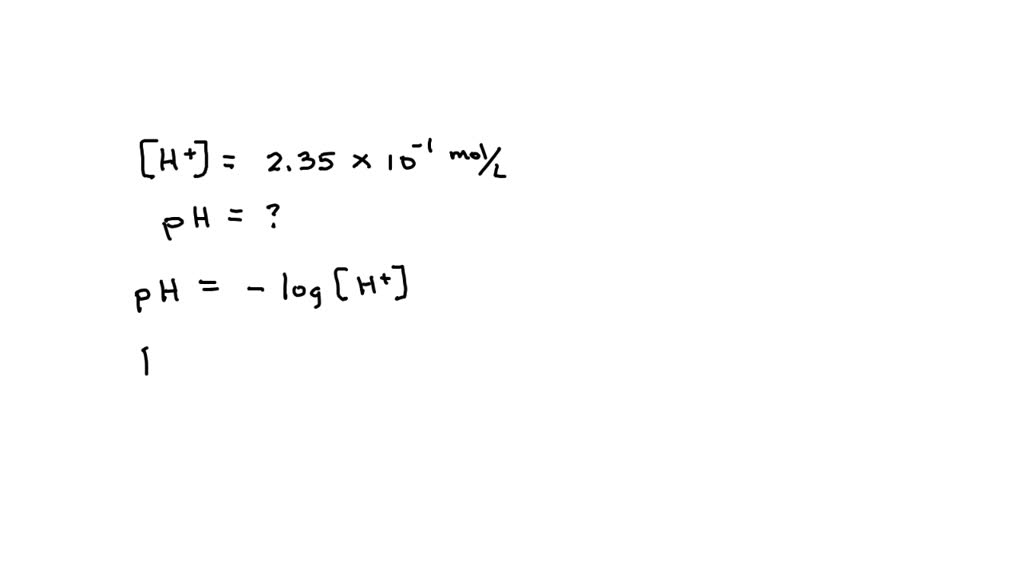
![Calculating [H+] from pH, Acids & Bases Tutorial - YouTube](https://i.ytimg.com/vi/bP-evPgNNUg/maxresdefault.jpg)



![SOLVED: The pH scale for acidity is defined by p = -log[H+], where H+ ...](https://cdn.numerade.com/ask_images/9a9653d07f19487eb2751645f28835fb.jpg)



![Calculating pH & pOH, [H+], [OH-], Acids & Bases CLEAR & SIMPLE - YouTube](https://i.ytimg.com/vi/eoM1nzYFy8k/maxresdefault.jpg)
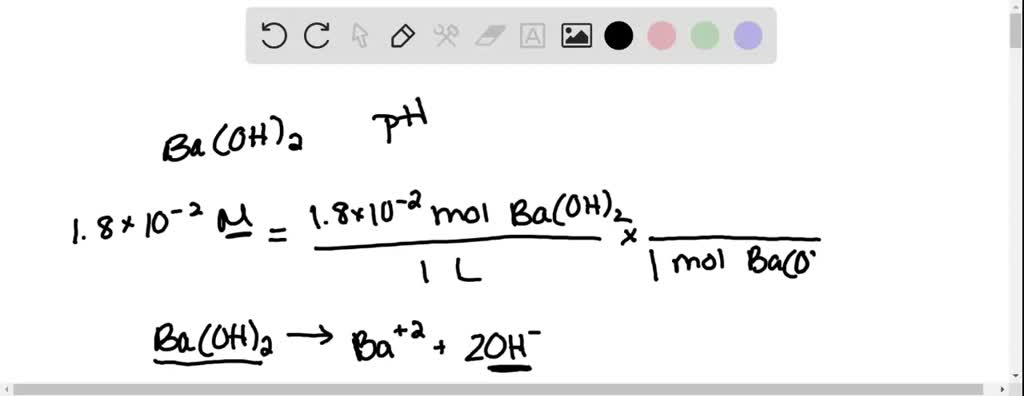
![Calculate the [H+] concentration for the following solution: a. pH = 3.45](https://cdn.numerade.com/ask_previews/eb4ab-202b-dd8d-3a6a-621415b612d1_large.jpg)
.PNG)
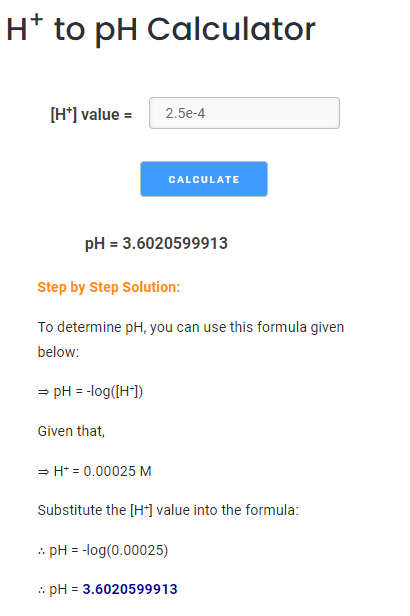



![Given [H+] or [OH-], Calculate pH & pOH - YouTube](https://i.ytimg.com/vi/ghIYaqo0Ycc/maxresdefault.jpg)
![pH of a solution is 3 calculate its [H+] ion concentration. #chemistry ...](https://i.ytimg.com/vi/VKRKpN3fkn4/oar2.jpg?sqp=-oaymwEkCJUDENAFSFqQAgHyq4qpAxMIARUAAAAAJQAAyEI9AICiQ3gB&rs=AOn4CLB6X8EOFhMOXVS-SjUhzq8zKGsZBA)


![Calculating pH, pOH, [H+], [H3O+], [OH-] of Acids and Bases - Practice ...](https://i.ytimg.com/vi/UiK37I159fc/maxresdefault.jpg)

![Solved 4. Calculate the [H+] concentration given each of the | Chegg.com](https://media.cheggcdn.com/media/a7a/a7a07342-5791-4205-8090-238e34dfda80/phpCVbarK)

![Solved Calculate [H+] and the pH of the following solutions. | Chegg.com](https://d2vlcm61l7u1fs.cloudfront.net/media/a4f/a4f063b0-ba07-4795-9cd8-580e949075c7/phphOs2vO.png)


![Solved Lab 15 F. CALCULATING PH, [H+], AND [OH-'J | Chegg.com](https://media.cheggcdn.com/study/839/8390c4ac-bd7f-4e38-9173-0b228425ec1a/image)
![SOLVED: The pH scale for acidity is defined by pH = log[H+], where [H+ ...](https://cdn.numerade.com/ask_images/a6d481cce0c9419387ec908ff7739db1.jpg)
![The pH scale for acidity is defined by pH = -log10[H+] where [H+] is ...](https://cdn.numerade.com/ask_images/5522d33e25844cc687bcdff93a334193.jpg)
![Solved Calculate the [H+] concentration given each of the | Chegg.com](https://media.cheggcdn.com/study/383/383210a4-6285-4524-9c93-1b17416f49ae/image)

![The pH scale for acidity is defined by pH = -log10 [H+] where [H+] is ...](https://cdn.numerade.com/ask_images/ce1cb2d5a1564f5090498becbe732a16.jpg)



![SOLVED: The pH scale for acidity is defined by pH = -log[H+], where [H+ ...](https://cdn.numerade.com/ask_images/4fe59e79394449bb845719ff93ab7a95.jpg)

![SOLVED: The pH scale for acidity is defined by pH = -log[H+] where [H+ ...](https://cdn.numerade.com/ask_images/f2767002ab994da9b0bf281dfafabb55.jpg)


![Using the equations: pH = -log [H+] and pH = 14 - pOH,...](https://cdn.numerade.com/ask_previews/dbf69eb9-458a-4bce-968f-95a2d4697109.gif)

![The pH scale for acidity is defined by pH = -log10 [H+] where [H+] is ...](https://cdn.numerade.com/ask_images/20666f6f23544a8c9fd39983cb950b6b.jpg)
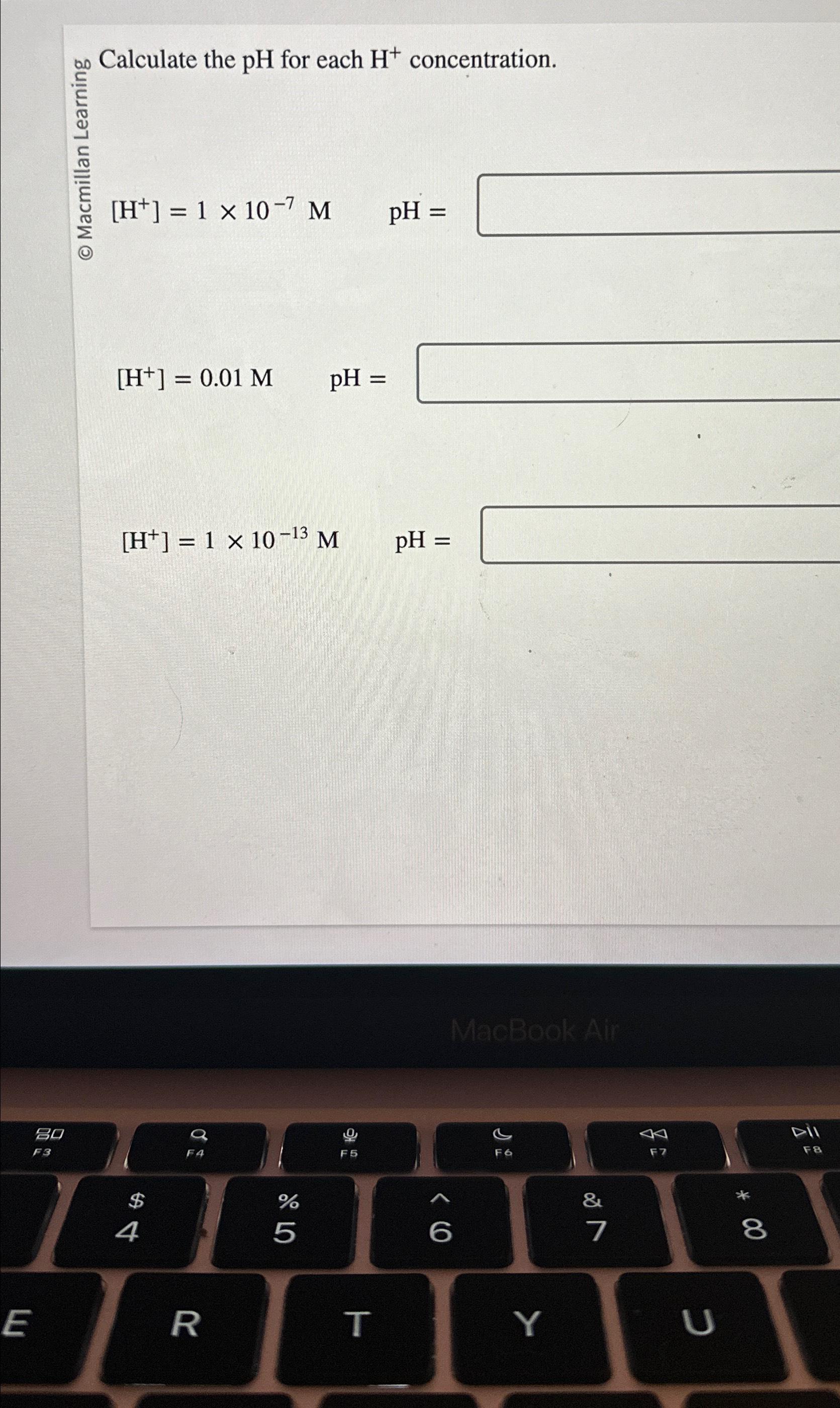
![SOLVED: The pH scale for acidity is defined by pH = -log[H+], where [H+ ...](https://cdn.numerade.com/ask_images/747cc4fb7dea4f9ab269f28b380d5724.jpg)
![The pH scale for acidity is defined by pH = -log10 [H+] where [H+] is ...](https://cdn.numerade.com/ask_images/510d8447b444484eafbbc492295c2de0.jpg)

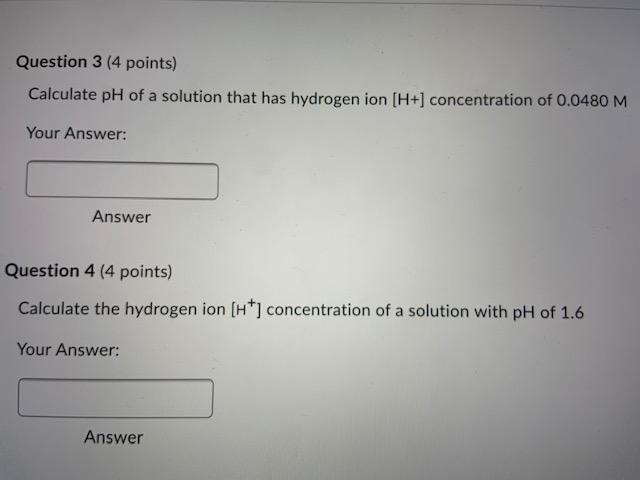

![pH Calculator - Calculate pH, pOH, and [H+] with Step-by-Step Solutions](https://images.miniwebtool.com/ph-calculator.png)

![Solved Calculate pH for each H^+ concentration: a) [H^+] = | Chegg.com](https://d2vlcm61l7u1fs.cloudfront.net/media/7d3/7d37f389-dd98-48c7-bac4-89893912504a/image)

![pH and [H+(aq)] | OCR A-Level Chemistry](https://www.chemistrystudent.com/ocr-a-level/5.1.3-acids-bases-and-buffers/images/h+ph.png)


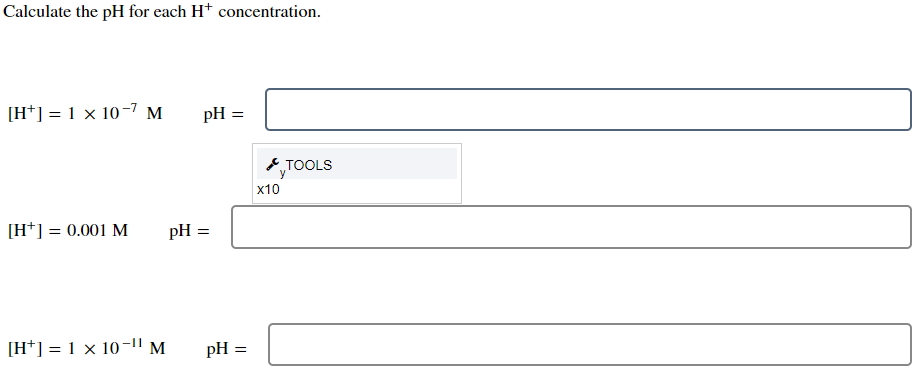







![SOLVED: Calculate the hydrogen ion concentration, [H+],for each of the ...](https://cdn.numerade.com/previews/f9af8115-1fb3-4081-864e-c69c0569cf8d_large.jpg)


![VIDEO solution: Calculate the pH, pOH, [H+] and [OH-] of a solution ...](https://cdn.numerade.com/ask_previews/a0de1f58-a1b8-448d-9e76-6b77083820b4_large.jpg)






![Calculations of pH, pOH, [H+] and [OH-]](https://www.sciencegeek.net/Chemistry/taters/graphics/pHSchematic.gif)